In a series of posts, we are going to talk about Mass Spectrometry.
- Introduction-The different configurations and the Electron Impact process
- What types of mass analyzers are there?
- What type of detectors are there?
- What types of analysis can be done?
- How do you read the output?
- How do they come to a qualitative measure using software?
- How do they quantitate the results?
- Do you need chromatography if you are using Mass Spectrometry?
- Other topics of interest about GC-MS
I am being very specific with my language here when the question posed is “How do THEY come to a qualitative measure using software?” and is not the question of “How SHOULD you come to a qualitative measure using software?”
Let’s look at how THEY do it first….
In this blog, we have posted on this particular topic before. Therefore, I would encourage you to review that post now, then come back to this post. Professor McLafferty of Cornell once wrote in his book Interpretation of Mass Spectra, “The mass spectrum shows the mass of the molecule and the masses of pieces from it. Thus the chemist does not have to learn anything new– the approach is similar to an arithmetic brain-teaser.”
Mass Spectrometry is only computer assisted pattern recognition
Now, presuming you have reviewed the above earlier post, we can look at some of the most amazing parts of MS work (at least to me) and see how really subjective it really is. It is truly open to interpretation.
When we just simply run the NIST spectral library searches, we have some issues of concern.
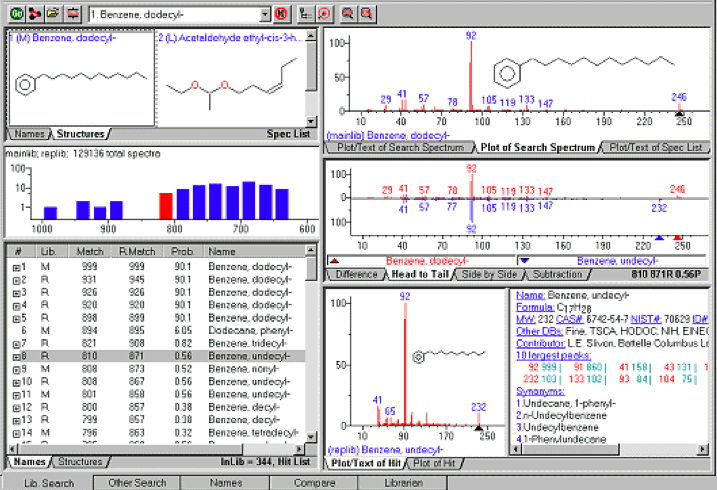
- The Graphic User Interface for GC-MS work
As it comes to pass that all that they do when it comes to the qualitative measure in GC-MS that is EI-based is perform computer assisted pattern recognition, then we need to be sure that the standard the unknown is compared against is from an unimpeachable source. Is it a traceable library of the spectra or is it local forced integration that is anecdotal in nature and therefore not independently adjudicated?
To try to solve this uniformity problem and to try to homogenize the standards, Professor Fred McLafferty of Cornell University in 1995 began to get his colleagues together and collect spectra. This resulted in the first EI-based mass spectrometry library that later was converted to what we now enjoy as the NIST/EPA/NIH Mass Spectral Library. The most current version of the EI Library in the NIST ’08 includes:
- mainlib (main EI MS library)=191,436
- replib (replicate spectra)=28,307
- nist_salts (EI Salt Library)=717
- nist_msms (MS/MS) Library=14,802 of which 3,898 are positive and 1,410 are negative
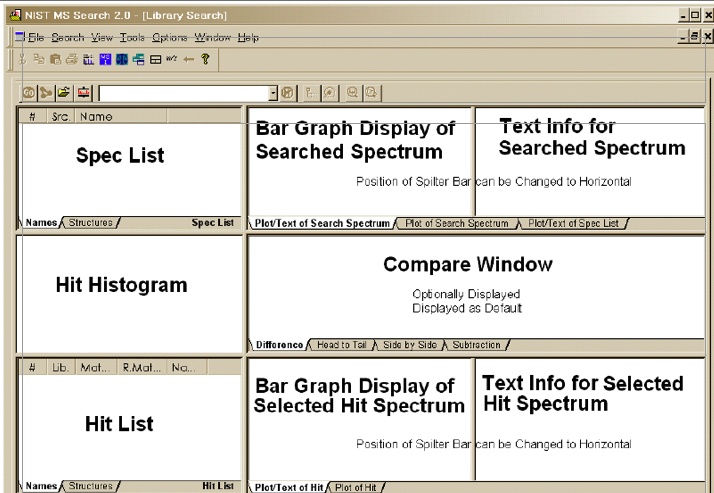
- A legend of the graphic user interface
On the graphic user interface as shown above, there are three very important places that we want to know about in order to judge the validity of this qualitative measure: the text info for the selected hit spectrum, the hit histogram, and the hit list.
All of these three key areas are affected by the particular type of search algorithm that is conducted. Is it an identity search or a similarity search?
- An Identity search is designed to find exact matches of the compound that produced the submitted spectrum and therefore presumes that the unknown compound is represented in the reference library.
- A “Similarity” search is optimized to find similar compounds and is intended for use when a compound cannot be identified by the “Identity” search.
What happens in the real world is that the analyst looks may look at the Hit Histogram and then the Hit List. They generally ignore the other screens and rarely look at the Text Info for the Selected Hit Spectrum, although it is perhaps the most important window.
- The Hit List has different columns of information. The Match Factor is an arbitrary unit number where a perfect match is 1000. It is the comparison between the unknown and the library (direct match). The probability value for a hit is derived assuming that the compound is represented by a spectrum in the libraries searched. It only employs the difference between adjacent hits in the hit list to get the relative probability that any hit in the hit list is correct. While many state scientists discount the probability value, it is arguably, the most “true” (correct) value that we can use to judge the value of the qualitative judgment call. Although there is no written criteria that is universal, it is thought that a match score of 950 or greater is considered an excellent match; 900-950 is a good match; 800-900 is a fair match; less than 800 is a very poor match. The Hit List can be thought of like a ranked top 100 list of compounds that the computer think the unknown is.
- The Hit Histogram is the number of hits vs. their Match Factors. It is displayed in the pane located just above the Hit List.
- The “Text Info for the Selected Hit Spectrum” will give us the name of the compound, its diagnostic ions, its CAS registry number, its NIST number (if it is a NIST traceable spectrum) and most importantly the source of where the standard comes from.
The NIST Spectral Library search is very popular. It is a shortcut to thinking through the fundamentals that mass fragmentation is based upon which is nothing more than good old fashioned acid-base chemistry. Exclusive reliance upon this simplified method of analysis as a means of identification can lead to an improper answer. There has always been an upstream-downstream problem with the NIST spectral library where over the years inferior spectra have made their way to the official NIST library. In fact, Professor Mclafferty noted in his book Interpretation of Mass Spectra “[O]ver the last decade approximately 60,000 errors have been corrected in the reference file [of the NIST library].” Just consider the below as an example:
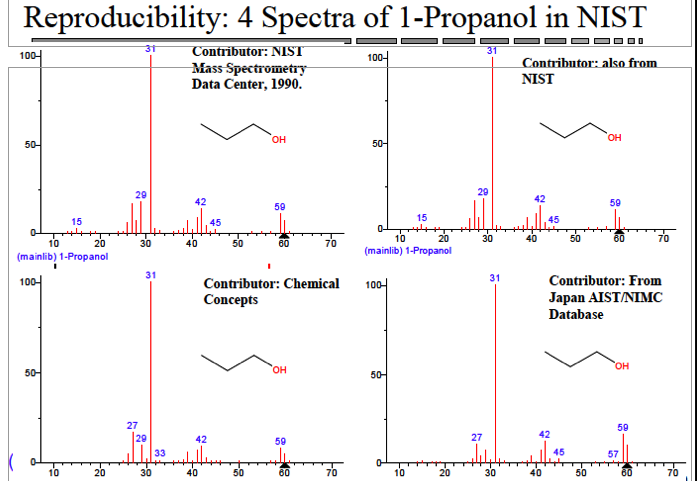
- Upstream-downstream problems exist in the NIST spectral library
At least one organization has published that simple reliance on any mass spectral library is insufficient in and of itself. SOFT/AAFS Forensic Toxicology Laboratory Guidelines 2006 version in 2006 in section 8.2.10 we find the following language:
8.2.10 In routine practice, interpretation of GC/MS-EI full scan mass spectra is performed by the instrument’s software as a semi-automated search against a commercial or user-compiled library. The quality of the match or “fit” may be aided by the factor that is generated, either as a ratio or percentage, where 1.0 or 100% are “perfect” matches. However, such “match factors” must be used as guides only and are not sufficiently reliable to be used as the final determinant of identification. Final review of a “library match” must be performed by a toxicologist with considerable experience in interpreting mass spectra; experience and critical judgement are essential. Interpretation, at a minimum, should be based on the following principles:
For a match to be considered “positive”, all of the major and diagnostic ions present in the known (reference) spectrum must be present in the “unknown”. Occasionally, ions that are in the reference spectra may be missing from the “unknown” due to the low overall abundance of the mass spectrum. If additional major ions are present in the “unknown” it is good practice to try to determine if the “extra” ions are from a co-eluting substance or “background” such as column bleed or diffusion pump oil. Examination of reconstructed ion chromatograms of the suspected co-eluting substance relative to major ions from the reference spectrum will help to determine this.
[Thank you to Dr. Stefan Rose, MD for pointing out the reference to the SOFT/AAFS guideline.]
Early in the same standard we find the following language:
8.2.9 Where mass spectrometry is used in selected ion monitoring mode for the identification of an analyte, whether as part of a quantitative procedure or not, the use of at least one qualifying ion for each analyte and internal standard, in addition to a primary ion for each, is strongly encouraged where possible. Commonly used acceptance criteria for ion ratios is ±20% relative to that of the corresponding control or calibrator. However, it is recognized that some ion ratios are concentration dependent and that comparison to a calibrator or control of similar concentration may be necessary, rather than an average for the entire calibration. Ion ratios for LC/MS assays may be more concentration and time dependent than for GC/MS and therefore acceptable ion ratio ranges of up to ±25% or 30% may be appropriate.
However, the ultimate question is: “How SHOULD we come to a qualitative measure using software?”
The answer is simple and old: Acid-base chemistry approach as articulated by Drs. Fred W. McLafferty and Frantisek Turecek in their book Interpretation of Mass Spectra. In this book they set out a “Standard Interpretation Procedure” which provides a useful and universal standard of how to use the powerful tool of Mass Spectrometry correctly to arrive at a qualitative measure. The “Standard Interpretation Procedure” is as follows:
- Study all available information (spectroscopic, chemical, sample history). Give explicit directions for obtaining spectrum. Verify m/v assignments.
- Using isotopic abundances where possible deduce the elemental composition of each peak in the spectrum; calculate rings plus double bonds.
- Test molecular ion identity; must be highest mass peak in spectrum, odd-electron ion, and give logical neutral losses. Check with CI or other soft ionization. (Don’t just rely on the EI-based GC-MS result)
- Mark “important” ions: odd-electron and those of highest abundance, highest mass, and/or highest mass in a group peaks.
- Study general appearance of spectrum; molecular stability, labile bonds.
- Postulate and rank possible structural assignments for:
(a) important low-mass ions series;
(b) important primary neutral fragments from M+ indicated by high-mass ions (loss of largest alkyl favored) plus those from secondary fragmentations indicated by CAD spectra;
(c) important characteristic ions.
- Postulate molecular structures; test against reference spectrum, against spectra of similar compounds, or against spectra predicted from mechanisms of ion decompositions.
Although there is not formally an eighth step Professor McLafferty reminds us that there is another important aspect to remember in all of this if we want to make sure that we are reporting out valid and high quality results with great confidence. He writes:
Remember that abundance values can vary by more than an order of magnitude between instruments; so you must measure a reference spectra of the postulated compound under the same instrumental conditions used with the unknown in order to have high confidence in the answer.
Later he writes:
“The most reliable match is obtained by running the unknown and reference mass spectra under closely identical experimental conditions on the same instrument.”
So even the “father” of the NIST library requires there to be verification by running reference materials to confirm spectrum are in fact repeatable under the specific conditions of the particular instrument. Dr. Marvin C. McMaster has plainly expressed the same notion in the following thoughts:
One of the problems with spectral library databases is that some of their structures are inaccurate or just plain wrong. The original interpretation of their structures may have been incorrect or mistakes may have been made in entering them. The previous Wiley/NIST spectral database with 225,000 compounds was thought to have up to 8% incorrect structures.
There are many aspects of GC-MS that can lead to erroneous identifications beyond this upstream-downstream problem such as:
- Were the tuning conditions used to prepare the reference standard in the library the same or different than the one that the unknown was run upon?
- The unknown and the reference spectra could have been run on a different type of mass spectrometer with different mass linearity. For example, some of the spectra in the library are so old that they were run on magnetic sector instruments rather than quadrupoles.
- The reference standard in the library may not have been from a pure compound. If the reference spectra in the library were run on impure compounds, then there will likely be additional fragment peaks due to the overlapping and the lack of resolution between compounds.
GC-MS work should be reserved to highly scientifically trained credentialed scientists who have a fundamental and strong understanding of chemistry, not someone who can simply follow a procedure, right double click and read a screen without giving any thought to it all.


